
Fill and finish services
Aseptic fill‑and‑finish is a critical final stage in the manufacture of therapeutic goods. This process requires specialised, validated equipment, robust process controls, and highly trained personnel to ensure accuracy, efficiency and full regulatory compliance.
At Pure Solutions, we deliver end‑to‑end sterile manufacturing supported by accredited and licensed facilities. Our operations are supported by comprehensive sterility assurance and quality control programs, including environmental monitoring, in‑process controls and sterility testing.
We are a licensed manufacturer of human and animal healthcare products, including sterile and non‑sterile (low‑bioburden) therapeutic goods. Our capabilities include formulation, fill‑and‑finish, labelling, packaging, storage, and release for supply.


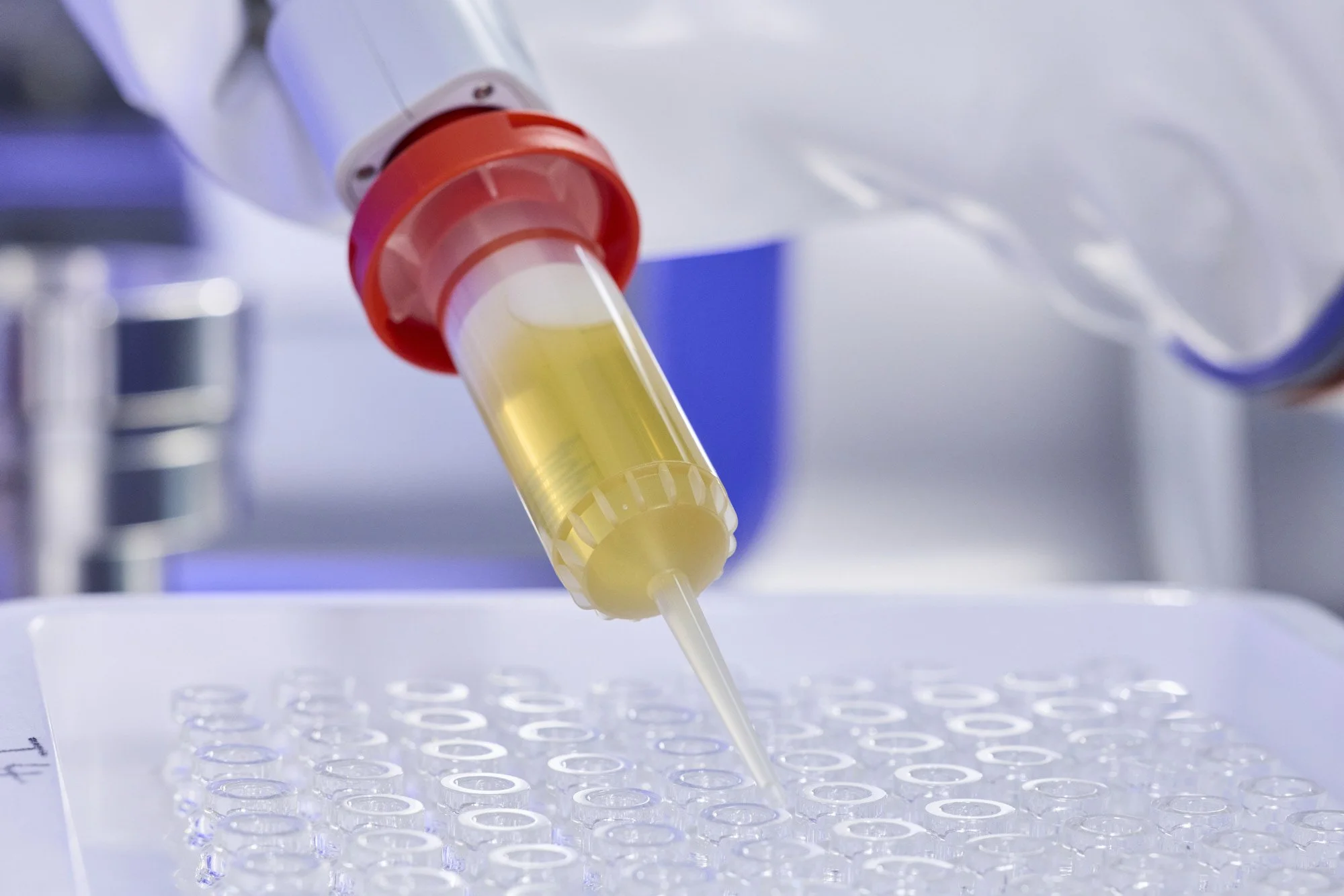
Grade A BioQuell Isolator technology with vaporised hydrogen peroxide (VHP)
Filling of vials, bags and droppers
High speed fully automated fill line
Integrated labelling line
Hand packing line

Labelling and packing
Aseptic manufacture of sterile product line
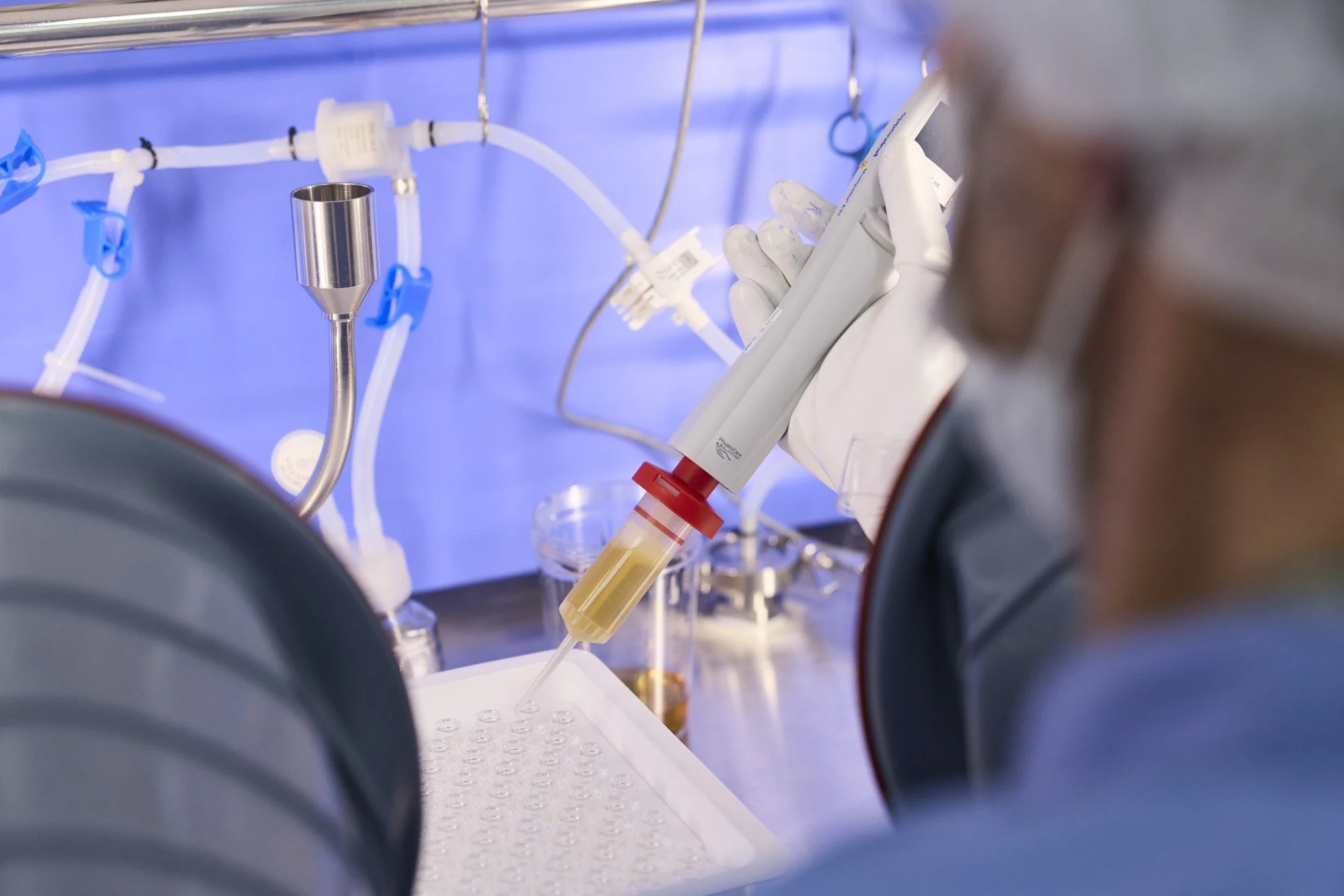
Non-sterile specialty line

Automatic labelling machine
Blinding Annex 13 compliant

Find out more

Comprehensive and advanced testing
Microbiological services
Download the Sample Analysis Form to submit your analysis request

